Fluorine
By Abigail Dobies
*Bolded word's definitions can be
found at the bottom of the page*
found at the bottom of the page*
General Information

My element is Fluorine. Fluorine's chemical symbol is F and it's atomic number is 9. This means that it has 9 protons, and 9 electrons. It's atomic mass is 19.00 and from this, you can figure out that it has 10 neutrons. Fluorine is a gas, and is in the non-metal element group. As you can see, it's pure color is a pale yellow.
Image Source: http://edtech2.boisestate.edu/mary
tsadi/images/vtourimages/fluorine_linkto.jpg
Image Source: http://edtech2.boisestate.edu/mary
tsadi/images/vtourimages/fluorine_linkto.jpg
A Fluorine Atom
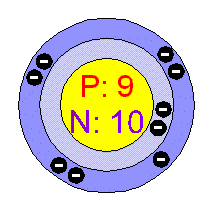
To the left is a demonstration of the protons (P), neutrons (N), and electrons (-) in a fluorine atom. As you can see, there are 9 protons, 10 neutrons, and if you count the (-) signs there are 9 electrons. Like explained above, since it's atomic number is 9, we know it has 9 protons and 9 electrons. Since fluorine's atomic mass is 19.00, we can figure out that it has 10 neutrons ( because it's mass, 19.00, minus either the number of protons/electrons equals the number of neutrons).
Image Source: http://www.chemicalelements.com/bohr/b0009.gif
Image Source: http://www.chemicalelements.com/bohr/b0009.gif
Why is Fluorine Important?
Fluorine is important because it is used in things that help prevent tooth decay! Without it, you would get cavities, your teeth would decay, and they would have plaque! This is extremely bad for you, which is why fluorine is so important. Fluorine is also important because we use fluorine EVERY DAY (for brushing teeth, mouthwash, and more)! Fluorine is also extremely important because it has become an essential part of drug discovery. About one fifth of all drug (medicines) contain a certain amount fluorine! Drugs (for helping people with diseases, medical problems, and more) are extremely important, which just shows how essential fluorine is.
(Fluorine is also very important for all of it's uses listed below).
(Fluorine is also very important for all of it's uses listed below).
Uses of Fluorine1) Fluorine is used in fluoride (at the dentist) which helps prevent dental cavities.
2) Toothpaste (sodium fluoride). 3) In some communities, part of fluorine is regularly added to drinking (tap) water (for the same reason as fluoride and toothpaste--to prevent plaque, cavities, and tooth decay) 4) One of fluorine's compounds, uranium hexafluoride, is used in processing nuclear fuels 5) Another compound of fluorine, Chlorofluorocarbons, used to be used for air conditioning, and refrigeration and cooling systems (fluorine was a part of Freons, which helped make the refrigerator cold), but later it was found that they were destroying the Earth's ozone layer, so it was banned. If they did not ban it, and the ozone layer was destroyed, we would all get burned, and die, which is exactly why they banned it! 6) It is used in rocket fuels since it is so reactive. 7) Hydrofluoric acid is used to etch (engrave) the glass in light bulbs and other products. 8) Fluoride is used in mouthwash (fluoride rinse) 9) Polytetrafluoroethylene (a compound containing fluorine) is used as a non-stick coating for cookware 10) Fluorine is used in many drugs, like anesthetics, which is a drug that causes a loss of sensation (puts you to sleep most often during surgery), and also analgesics, which are pain killers. |
Physical Properties of Fluorine1) The melting point of Fluorine is -219.62°C
2) The boiling point of Fluorine is -188.12°C 3) Fluorine has quite a strong odor 4) As said above, fluorine's pure color is a pale yellow 5) Fluorine has a density of about 1.696 grams per liter at standard temperature and pressure (STP). |
Cool Facts about Fluorine1) Fluorine is the most reactive of all of the elements!
2) Fluorine reacts with all of the elements except (directly) oxygen, helium, krypton, and neon. 3) Fluorine is the lightest of the Halogens 4) Fluorine is the most electronegative element, which means it will "grab" electrons from other atoms very easily. This is why if you take fluorine and put it with any other element in the Periodic Table (except for neon, helium, oxygen, and krypton) there will be a reaction. 5) Fluorine causes severe chemical burns on contact with skin! 6) While working with fluorine, some scientists have experienced fatalities and blindings! |
Pictures of Fluorine's Uses:
Toothpaste/Fluoride Refrigerators Etching Glass
Mouthwash Some Drinking Water Rocket Fuels
Non-stick Coating Drugs (Medicine)
Image Sources:Toothpaste/Fluoride: http://periodicnetwork2011tag.pbworks.com/f/1320783170/Profile_Fluorine.jpg
Refrigerators: http://www.geappliances.com/appliances/refrigerators/images/thumb_bottom_freezer_refrigerators.jpg
Etching Glass: http://img1.etsystatic.com/000/0/6409076/il_fullxfull.259612653.jpg
Mouthwash: http://bestmouthwash.org/wp-content/uploads/2012/02/best-mouthwash-ACT-Kids-Anticavity-Fluoride-Mouthwash.jpg
Some Drinking Water: http://www.globalresearch.ca/wp-content/uploads/2013/02/water-fluoridation-1.jpg
Rocket Fuels: http://www.howitworksdaily.com/wp-content/uploads/2012/05/Rocket_launch.jpg
Non-Stick Coating: http://www.meijer.com/assets/product_images/styles/xlarge/1001029_051500117392_A_400.jpg
Drugs (Medicine): http://static3.depositphotos.com/1005091/226/v/950/depositphotos_2261092-Two-cartoon-doctors.jpg
Refrigerators: http://www.geappliances.com/appliances/refrigerators/images/thumb_bottom_freezer_refrigerators.jpg
Etching Glass: http://img1.etsystatic.com/000/0/6409076/il_fullxfull.259612653.jpg
Mouthwash: http://bestmouthwash.org/wp-content/uploads/2012/02/best-mouthwash-ACT-Kids-Anticavity-Fluoride-Mouthwash.jpg
Some Drinking Water: http://www.globalresearch.ca/wp-content/uploads/2013/02/water-fluoridation-1.jpg
Rocket Fuels: http://www.howitworksdaily.com/wp-content/uploads/2012/05/Rocket_launch.jpg
Non-Stick Coating: http://www.meijer.com/assets/product_images/styles/xlarge/1001029_051500117392_A_400.jpg
Drugs (Medicine): http://static3.depositphotos.com/1005091/226/v/950/depositphotos_2261092-Two-cartoon-doctors.jpg
Pronunciation/Backround:
Pronunciation:
Fluorine is pronounced: FLOO-uh-reen.
Backround:
In 1813, Sir Humphry Davy named the element. Fluorine was named from the mineral fluorspar, who's name was given by Georgius Agricola in 1546. 'Fluor' in fluorspar came from the Latin word 'fluere', which meant 'to flow' because it allowed metals to flow. So, basically the name 'fluorine' came from the 'fluor' in fluorspar (below).
Fluorine is pronounced: FLOO-uh-reen.
Backround:
In 1813, Sir Humphry Davy named the element. Fluorine was named from the mineral fluorspar, who's name was given by Georgius Agricola in 1546. 'Fluor' in fluorspar came from the Latin word 'fluere', which meant 'to flow' because it allowed metals to flow. So, basically the name 'fluorine' came from the 'fluor' in fluorspar (below).
The Discovery of Fluorine

Henri Moissan
(1852-1907)
Fluorine was first described by Georgius Agricola in 1529. Then, in 1813, a chemist named Sir Humphrey Davy tried to extract fluorine, but was not successful. In 1869, George Gore was able to produce a small amount of fluorine, but since he didn't know much about fluorine, it quickly combined with another element (hydrogen). Finally, fluorine was successfully isolated by Henri Moissan, a French chemist, in 1886. He performed electrolysis, which meant using electric currents to make a reaction. It seperates elements from naturally occuring things, and in this case, fluorspar. For Moissan's great achievement, he won a Nobel Prize in chemistry in 1906.
Image Source: http://upload.wikimedia.org/wikipedia/commons/3/3d/Henri_Moissan_1896.jpg
Image Source: http://upload.wikimedia.org/wikipedia/commons/3/3d/Henri_Moissan_1896.jpg
Fluorine Video...
Video Source: http://www.youtube.com/watch?v=Efpk01iixkI
The Reaction of Cesium and Fluorine-for the FIRST time on camera!
Video Source: http://www.youtube.com/watch?feature=player_embedded&v=TLOFaWdPxB0
Quick Information
Chemical Name: Fluorine
Chemical Symbol: F
Atomic Number: 9
Atomic Mass: 19.00
Protons: 9
Electrons: 9
Neutrons: 10
Color: Pale Yellow
Non-metal, metalloid, or metal: Non-metal
Solid, Gas, Liquid, or Synthetic: Gas
Half-life (of fluorine with an atomic mass of 19.00): Stable
Group: 17
Period: 2
Chemical Symbol: F
Atomic Number: 9
Atomic Mass: 19.00
Protons: 9
Electrons: 9
Neutrons: 10
Color: Pale Yellow
Non-metal, metalloid, or metal: Non-metal
Solid, Gas, Liquid, or Synthetic: Gas
Half-life (of fluorine with an atomic mass of 19.00): Stable
Group: 17
Period: 2
Definitions
Ozone layer: The ozone layer is a layer in the Earth's atmosphere which absorbs (blocks) 97-99% of the sun's ultraviolet radiation (heat from the sun).
Electronegative: Being an electronegative element means the element will "grab" electrons from other atoms very easily. This is why if you take fluorine (an electronegative element) and put it with any other element in the Periodic Table (except for neon, helium, oxygen, and krypton) there will be a reaction.
Halogen: A halogen is one of the groups in the Periodic Table of Elements. The halogens are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At), which all have similarities like they are all non-metals, are very reactive, and have 7 electrons in their outer shells (in the atom). Halogen comes from the Greek words 'halos' which means 'salt', and 'genos' which means 'produce'. Therefore, halogens can produce salt, and halogen also means "salt-former", and compounds containing (that have) halogens are called "salts" (can form salts).
Group: On the Periodic Table of Elements, the columns are called groups. Elements within a group share similar properties. Fluorine is in the Halogen group (group 17).
Period: On the Periodic Table of Elements, the rows (across) are called periods. Atomic number increases as you move across a period.
Directly: This means that it is not a compound of fluorine reacting (or an extraction), just elemental fluorine doesn't react with these 3 elements.
Electronegative: Being an electronegative element means the element will "grab" electrons from other atoms very easily. This is why if you take fluorine (an electronegative element) and put it with any other element in the Periodic Table (except for neon, helium, oxygen, and krypton) there will be a reaction.
Halogen: A halogen is one of the groups in the Periodic Table of Elements. The halogens are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At), which all have similarities like they are all non-metals, are very reactive, and have 7 electrons in their outer shells (in the atom). Halogen comes from the Greek words 'halos' which means 'salt', and 'genos' which means 'produce'. Therefore, halogens can produce salt, and halogen also means "salt-former", and compounds containing (that have) halogens are called "salts" (can form salts).
Group: On the Periodic Table of Elements, the columns are called groups. Elements within a group share similar properties. Fluorine is in the Halogen group (group 17).
Period: On the Periodic Table of Elements, the rows (across) are called periods. Atomic number increases as you move across a period.
Directly: This means that it is not a compound of fluorine reacting (or an extraction), just elemental fluorine doesn't react with these 3 elements.
Bibliography
Works Cited
(Websites)
1)
Bentor, Yinon. "Chemical Elements.com - Fluorine (F)." Chemical Elements.com. Yinon Bentor, 1996. Web. 20 Mar. 2013.
2)
"Fluorine Uses." Buzzle.com. Buzzle, 2000. Web. 23 Mar. 2013.
3)
Stewart, Doug. "Fluorine Element Facts." Chemicool.com. David D. Hsu, 1996. Web. 14 Mar. 2013.
4)
Winter, Mark J. "Fluorine." Periodic Table of the Elements by WebElements.com Dr. Mark Winter, Sept. 1993. Web. 23 Mar. 2013.
(Encyclopedia)
5)
The World Book Encyclopedia. Volume 7. 2009. Print.
(Non-fiction Book)
6)
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Vol. 14. Danbury, CT: Grolier Educational, 1996. Print. Chlorine, Fluorine, Bromine, and Iodine.
(Websites)
1)
Bentor, Yinon. "Chemical Elements.com - Fluorine (F)." Chemical Elements.com. Yinon Bentor, 1996. Web. 20 Mar. 2013.
2)
"Fluorine Uses." Buzzle.com. Buzzle, 2000. Web. 23 Mar. 2013.
3)
Stewart, Doug. "Fluorine Element Facts." Chemicool.com. David D. Hsu, 1996. Web. 14 Mar. 2013.
4)
Winter, Mark J. "Fluorine." Periodic Table of the Elements by WebElements.com Dr. Mark Winter, Sept. 1993. Web. 23 Mar. 2013.
(Encyclopedia)
5)
The World Book Encyclopedia. Volume 7. 2009. Print.
(Non-fiction Book)
6)
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Vol. 14. Danbury, CT: Grolier Educational, 1996. Print. Chlorine, Fluorine, Bromine, and Iodine.



