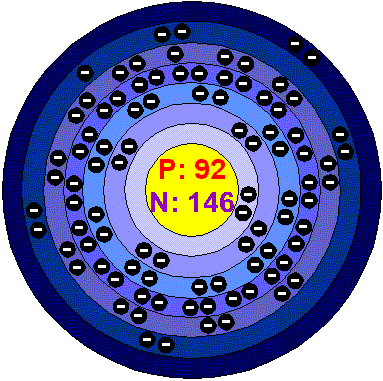
Uranium U
Basic Facts
- Chemical name: Uranium
-
- Chemical Symbol: U
-
- Atomic Number: 92
-
- Atomic Mass: 238.03
-
- Solid,Liquid,Gas, or Synthetic: Natural Solid
-
- Color: Silvery Gray
-
- Element Group Name: Metal
-
- Radioactive: Radioactive
-
- Melting Point: 2069.6 F
- Boiling Point: 6904.4 F
http://biobook.nerinxhs.org/bb/cells/biochemistry/424px-Uranium.svg.png
Discovery Uranium was discovered by Martin Klaproth in 1789 by isolating Uranium inside the mineral Pitchblende. Before this discovery, Martin Klaproth thought that Pitchblende was made of Iron and Zinc, But they found a new element inside of it and named it after the Planet Uranus. But what Martin Klaproth discovered wasn't pure Uranium it was Uranium oxide. Eugene Melchior Peligot produced the first pure Uranium from Uranium oxide in 1841.
http://photos.aip.org/history/Thumbnails/klaproth_martin_a1.jpg
http://upload.wikimedia.org/wikipedia/commons/thumb/e/ed/Eugene_peligot.jpg/220px-Eugene_peligot.jpg
http://farm6.static.flickr.com/5179/5399289753_fb4c990060.jpg
Physical Properties of Uranium
- Heavy
- Lustrous
- Softer then steel.
- When it is exposed to air, it gets coated in a layer of oxygen.
- It is also radioactive.
- It isn't hard enough to scratch glass.
- Acids will dissolve Uranium
- Uranium is toxic to consume.
http://www.chemicalelements.com/bohr/b0092.gif
http://macaulay.cuny.edu/eportfolios/bernstein08/files/2008/12/atremical.jpg
http://upload.wikimedia.org/wikipedia/commons/thumb/d/d4/Yellowcake.jpg/230px-Yellowcake.jpg
Why is Uranium important?
Uranium is an important element because it is used in the military for atomic bombs, tank armor, and high penetration tank shells. Uranium is used in nuclear reactors. Besides the military and nuclear reactors, it is used as weights for yachts and airplanes. Uranium is also used in smoke detectors and even in dating the age of the earth.
http://www.mindfully.org/Nucs/2005/Depleted-Uranium-Ethics9may05p15.jpg
http://www.chemcases.com/images/9%20-%20into.jpg
http://www.bbc.co.uk/schools/gcsebitesize/science/images/earth1.jpg
Where is Uranium Used?
- Uranium is used as fuel for nuclear reactors
- In ancient times uranium oxide was used to produce yellow colored glaze and was used to color pottery to kitchen tiles.
- Depleted Uranium is also used for high penetration tank shells and tank armor.
- Depleted Uranium {DU has about 60 percent radioactivety of Natural Uranium} of is used to shield things from radiation
- Used in atomic bombs
- Since it has a long half life it is used to date the age of the earth
- Used in ballasts in yachts and airplanes.
- Used in the chemicals for photography
- The radioisotopes of Uranium is used in smoke detectors.
- It is also used as poison
http://0.tqn.com/d/chemistry/1/0/p/9/1/uranium-glass-fluorescence.jpg
http://www.eoearth.org/files/122801_122900/122879/150px-Smoke-detector.gif
http://www.pioneerphotogallery.com/images/camera.gif
http://www.heartsupport.com/wp-content/uploads/2012/06/poison_bottle-1.jpg
Cool Facts!
- U-230 has a half life of 20.8 days
- U-231 has a half life of 4.2 days
- U-232 has a half life of 70 years
- U -233 has a half life of 159000 years
- U-234 has a half life of 247000 years
- U-235 has a half life of 70004e8 years
- U -236 has a half life of 234e7 years
- U-237 has a half life of 6.75 days
- U-238 has a half life of 447e9 years
- U-239 has a half life of 23.5 minutes
- U-240 has a half life of 14.1 hours
- By 2011 the price of Uranium was 51 dollars per pound
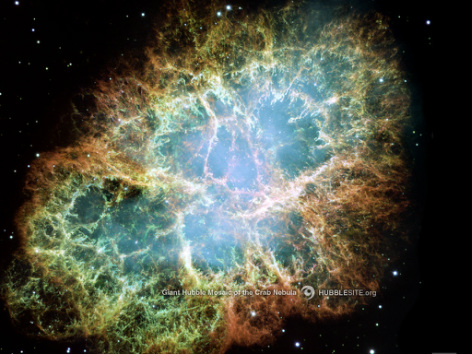
- Uranium is naturally created in Supernovas
http://ut-images.s3.amazonaws.com/wp-content/uploads/2009/10/crabnebulawallpaper.jpg
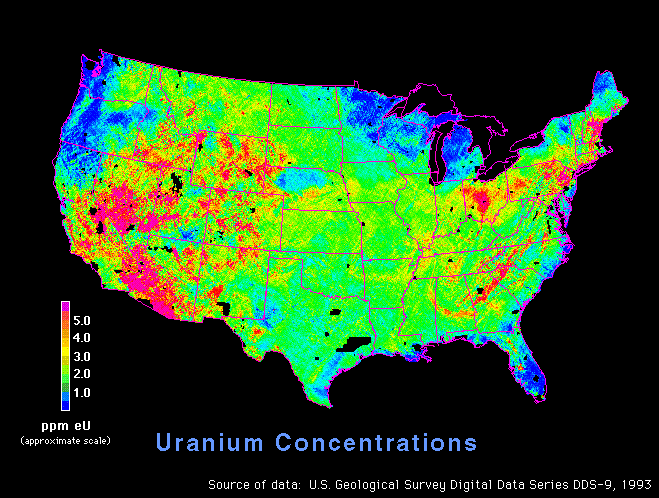
http://energy.cr.usgs.gov/radon/usaeu.gif
http://media.salon.com/2012/07/money_rect.jpg
Q and A
Q: Was there really a kid who carried Uranium ore to school by accident?
A: Not knowing if this is true there might of been a 12 year old in 1970 who carried in his pocket Uranium ore to school.
Q: Is Uranium the heaviest natural element known to us?
A: Uranium is the heaviest natural element.
Q:If you touch Uranium with your bare hands what will happen?
A: Nothing
Q: Are all the isotopes of Uranium radioactive?
A: All of them are radioactive
Q:How many pounds of Uranium were in the Hiroshima atomic bomb?
A: 141 pounds
Q: Is there an element that is more radioactive than Uranium
A: Radium and Polonium are way more radioactive then Uranium
Q:Does Uranium contain a lot of energy?
A: Yes, in fact 3 million pounds of coal is equal to 1 pound of Uranium
http://periodictable.com/Samples/092.19/s9s.JPG
http://periodictable.com/Samples/088.15/s9s.JPG
http://periodictable.com/Samples/084.8/s9s.JPG
This video shows the extreme destruction of Uranium. It also shows why to never mess with it!
http://www.youtube.com/watch?v=NpbCZ8QRpEg
This Video is about finding Uranium in Nature
http://www.youtube.com/watch?v=v9DsOyWsNp0
Bibliography
Works Cited
Bentor, Yinon. "Chemical Elements.com - An Interactive Periodic Table of the Elements." Chemical Elements.com - An Interactive Periodic Table of the Elements. Yinon Bentor, 2012. Web. 13 Mar. 2013.
Helminstein, Annie. "Periodic Table of the Elements." Periodic Table. About.com, 2004. Web. 13 Mar. 2013.
Hsu, David. "Alphabetical Elements List." Chemicool. David Hsu, 2013. Web. 13 Mar. 2013.
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. Print.
Pagan, Carmen. "Http://school.eb.com/comptons." Http://school.eb.com/comptons. Encyclopædia Britannica, 2013. Web. 18 Mar. 2013.
Rader, Andrew. "Periodic Table and the Elements." Chem4Kids.com: Elements & Periodic Table. Www.chem4kids.com, 2012. Web. 13 Mar. 2013.
Student Planner. 2012. Assignment Notebook.
Winter, Mark. "Home of the Periodic Table." Periodic Table of the Elements by WebElements. Mark Winter, 1993. Web. 13 Mar. 2013.
Bentor, Yinon. "Chemical Elements.com - An Interactive Periodic Table of the Elements." Chemical Elements.com - An Interactive Periodic Table of the Elements. Yinon Bentor, 2012. Web. 13 Mar. 2013.
Helminstein, Annie. "Periodic Table of the Elements." Periodic Table. About.com, 2004. Web. 13 Mar. 2013.
Hsu, David. "Alphabetical Elements List." Chemicool. David Hsu, 2013. Web. 13 Mar. 2013.
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. Print.
Pagan, Carmen. "Http://school.eb.com/comptons." Http://school.eb.com/comptons. Encyclopædia Britannica, 2013. Web. 18 Mar. 2013.
Rader, Andrew. "Periodic Table and the Elements." Chem4Kids.com: Elements & Periodic Table. Www.chem4kids.com, 2012. Web. 13 Mar. 2013.
Student Planner. 2012. Assignment Notebook.
Winter, Mark. "Home of the Periodic Table." Periodic Table of the Elements by WebElements. Mark Winter, 1993. Web. 13 Mar. 2013.