Zinc
By: Kelley Manley
*bolded words mean you can find there definitions at the bottom

Zinc was first discovered in 1746. It was discovered in Germany by Andreas Marggraf. He discovered it by heating a mixture of calamine, ore, and carbon in a closed tube to create zinc. Zinc is a solid, soft metal with a shiny surface, and started out as a gray metal, but when polished, it turn bluish white. Zinc is very important in industry because it has the ability to work well with other metals.
This is a picture of Andreas Maggraf
http://upload.wikimedia.org/wikipedia/commons/8/85/Andreas_Sigismund_Marggraf-flip.jpg
Zinc is pronounced "zingk"
This is a picture of Andreas Maggraf
http://upload.wikimedia.org/wikipedia/commons/8/85/Andreas_Sigismund_Marggraf-flip.jpg
Zinc is pronounced "zingk"
Where zinc is used or found:
Interesting facts:
- Brass-35% of zinc
- copper-65% of zinc
- alloys
- pills
- iron
- steel
- roof gutters
- electric batteries
Interesting facts:
- Is used for grooming dogs
- Good for bodies immune system
- Two thousandths of one percent of the earths crust
- Zinc has been used since prehistoric times
- Without zinc no one could survive
- If you dont have enough zinc in your body, you cant smell anything
ZINC!!!!!!!
Basics
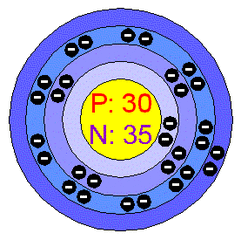
Element symbol: Zn
Atomic number: 30
Atomic mass: 65.39
Melting point: 419.58 °C
Boiling point: 907°C
Radioactive?: no
Group name: metal
Neutrons: 35
Protons: 30
Electrons: 30
http://www.chemicalelements.com/bohr/b0030.gif
Element symbol: Zn
Atomic number: 30
Atomic mass: 65.39
Melting point: 419.58 °C
Boiling point: 907°C
Radioactive?: no
Group name: metal
Neutrons: 35
Protons: 30
Electrons: 30
http://www.chemicalelements.com/bohr/b0030.gif
Important Words
*Alloys: a metal made by combining two or more metallic elements
*Metallic Element: several chemical elements that conduct heat or electricity
*Boiling Point: When the liquid changes into vapor
More Information:
*Alloys: a metal made by combining two or more metallic elements
*Metallic Element: several chemical elements that conduct heat or electricity
*Boiling Point: When the liquid changes into vapor
More Information:
Why did I pick this element?
I picked this element because I didn't know a lot about Zinc and I didn't really know how it was helpful in our daily lives.
Bibliography:
Works Cited
Bentor, Yinon. "Chemical Elements.com - Zinc (Zn)." Chemical Elements.com - Zinc (Zn). Yinon Bentor, 2012. Web. 24 Mar. 2013.
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. Print.
Robertson, Murray. "Zinc." - Element Information, Properties and Uses. Murray Robertson, 2011. Web. 24 Mar. 2013.
The World Book Encyclopedia. Chicago, IL: World Book, 2009. Print.
The World Book Student Discovery Encyclopedia. Chicago: World Book, 2005. Print.
"Zinc Element Facts." Chemicool. Chemicool, 2013. Web. 24 Mar. 2013.
