Titanium
By John Moore S3
You are one of the lucky people who get to learn about the universe's greatest element Titianium!!!! Not only it is one of the most commonly found elements in the crust of the earth, and is very strong not to mention the light weight too....
Origin
Titanium was discovered in 1791 by a scientist named Reverend William Gregor who was also a geolgist. Gregor was walking by and discovered some magnetic black sand that looked like gunpowder, by a stream near the parish of mannacan in Cornwall, England. (We call this sand now ilmenite) Gregor took some samples of it, and went to analyze. When he did, he discovered that it was magnetite (Fe3O4) and the rather oxide of new metal, which he described it as "reddish brown calx". This "calx" dissolved in sulfric acid and turned purple when reduced with iron, tin, or zinc. Gregor decided that he was dealing with a new metal. Gregor named the new metal "manaccanite" in honor of the parish of mannacan. Nothing really happens to our story until 1797 when german chemist Martin Klaproth discovered the same metal that Gregor found, but he named it Titanium. When Klaproth analysed it in 1797, Klaproth also read Gregor's account from 1791 about his discovery. He realized that the red oxide he found in his Titanium was in fact the same red oxide that was in mannacan. So both their elements were the same! Even though Gregor might have beat Klaproth to the element, scientists liked the name Titanium better. So that is how Titanium was what it is!
Here is a picture of Reverend William Gregor, the true discoverer of the element Titanium.
Picture Url: http://farm6.staticflickr.com/5169/5330173121_f5356b7681_z.jpg
Quick Facts

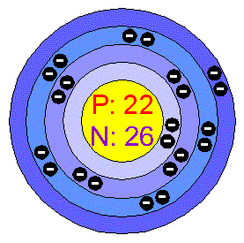
- Titanuim's chemical symbol is (Ti)
- It's atomic number is 22 that means it has 22 protons and electrons and 26 neutrons
- It's atomic mass is 47.87
- It is a Solid
- It has a silvery, chromey color
- It's element group is with the metals
- Titanium is stronger than steel and is 42% lighter
- The density of Titanium is 4.5 grams per cubic centimeter.
- The melting point of Titanium is at 3,304 degrees farenheit
- Titanium is metallic (Metal)
- Titanium is the 9th most abundant metal on the earth
- Titanium is a transition metal
Image Url http://pss.scdsb.on.ca/Departments/Science/
Titanuim's Importance
I think titanuim is important because it builds around our everyday lives, also it's there for you when you need it. What I mean by that is let's say cars for example if you accidentaly get trapped in quicksand and they have a device that can only lift a certain amount of weight. If you have titanium in your car, your in luck because of titanium being stronger than steel and 42% lighter you can have a modern car also with the density. Also say if you have a ship. Titanium has great resistance to salt water, so can propel right through it. But if you build with steel you have the possibility after awhile of the ship sinking because of the saltwater "eating away" at it. Also it is important because used as the compound Titanium dioxide (TiO2) a substance with an intense white color which it is used as a pigment in paper, paint and plastics those objects are essential in our society.
Physical Properties/ Characteristics
The properties and characteristics of pure titanium is a light, silvery-white, hard, lustrous metal. It has excellent strength and corrosion resistance and also has a high strength to weight ratio. Titanium’s corrosion rate is so low that if you put titanium for 4000 years in seawater, the corrosion would only have penetrated the metal to the thickness of a thin sheet of paper! At high temperatures titanium burns in air, but strangely titanium also burns in pure nitrogen. Titanium is ductile and is malleable when heated. Also and did you know Titanium metal is considered to be non-toxic, But as metal shavings, or powder, it is a considerable fire hazard. Titanium chlorides are also corrosive.
Everyday objects that contain Titanium

A decent amount of objects in our everyday lives contain Titanuim, in this passage I will show you that wide variety of objects.
1) Aerospace ( enginner of aircraft and space vehicles)
2) Hulls of ships also somethimes the propeller too. Basically any structure close or in the sea
3) Joint replacement implants, such as the ball and hip socket hip joint.
4) White paint
5) Food coloring, toothpaste, elastics, and sunscreen
6) Aicraft and Automobiles
7) Drill bits, bicycles, golf clubs, watches, and laptop computers
8) Military Tanks
Those are some of the everyday objects that increase capibilites because of titanium and make it more easier and more enjoable also. After this presantation maybe I will have pursuaded you to get an object containing Titanium inside of it!
Image Url: http://www.hdwallpapersfan.com/wp-content/uploads/2013/02/Aircraft-electrical-system.jpg
1) Aerospace ( enginner of aircraft and space vehicles)
2) Hulls of ships also somethimes the propeller too. Basically any structure close or in the sea
3) Joint replacement implants, such as the ball and hip socket hip joint.
4) White paint
5) Food coloring, toothpaste, elastics, and sunscreen
6) Aicraft and Automobiles
7) Drill bits, bicycles, golf clubs, watches, and laptop computers
8) Military Tanks
Those are some of the everyday objects that increase capibilites because of titanium and make it more easier and more enjoable also. After this presantation maybe I will have pursuaded you to get an object containing Titanium inside of it!
Image Url: http://www.hdwallpapersfan.com/wp-content/uploads/2013/02/Aircraft-electrical-system.jpg
Did you Know?
Another useful compound of Titanium is Titanium tetrachloride (TiCl4), a colorless liquid. When exposed to moist air, it forms a dense white cloud of small Titanium dioxide particles. The U.S navy used Titanium tetrachloride during world war 2 to make smokescreens when it was necessary to block a potential target from view.
Interesting facts
1) The annual production of Titanium dioxide in the U.S is approximately 1 million tons.
2) Titanium makes up about 0.57% of the earth's crust
3) Titanium is the ninth most abundant element on Earth
4) Since titanium does not react within the human body, it is used to create artificial hips, pins for setting bones and for other biological implants.
5) Some plants in the ocean might have a trace of Titanium in them.
Image Url: http://www.titanexport.com/img/tio2-eng.gif
2) Titanium makes up about 0.57% of the earth's crust
3) Titanium is the ninth most abundant element on Earth
4) Since titanium does not react within the human body, it is used to create artificial hips, pins for setting bones and for other biological implants.
5) Some plants in the ocean might have a trace of Titanium in them.
Image Url: http://www.titanexport.com/img/tio2-eng.gif
Story about Titanium
In the USA, the Department of Defense realized the strategic importance of the metal and supported early efforts of commercialization Throughout the period of the Cold War, titanium was considered a Strategic Material by the U.S. government, and a large stockpile of titanium sponge was maintained by the Defense National Stockpile Center, which was finally depleted in the 2000s. Today, the world's largest producer, Russian-based Vsmpo-Avisma, is estimated to account for about 29% of the world market share.
Isotpes
|
Isotpes
Ti-44 Ti-45 Ti-46 through Ti-50 Ti-51 |
Half Life
52 years 3.07 hours Stable 5.76 minutes |
What does that mean?
Oxide - An oxide is a chemical compound that contains at least one oxygen atom and one other element.
Vsmpo-Avisma - The world's largest titanium producer.
Ductile - is a solid material's ability to deform under tensile stress . Isotope - Atoms that have the same number of protons, but a different number of neutrons
Half life - is the time required for any quantity to fall to half its value as measured at the beginning of the time period.
Corrosion - The gradual destruction of a metal Pigment- a dry substance, usually reduced to particles, which can become paint, ink, etc. when suspended in a liquid vehicle.
Vsmpo-Avisma - The world's largest titanium producer.
Ductile - is a solid material's ability to deform under tensile stress . Isotope - Atoms that have the same number of protons, but a different number of neutrons
Half life - is the time required for any quantity to fall to half its value as measured at the beginning of the time period.
Corrosion - The gradual destruction of a metal Pigment- a dry substance, usually reduced to particles, which can become paint, ink, etc. when suspended in a liquid vehicle.
|
This is the location where Titanium was first discovered, by Reverend William Gregor. By the stream where the Parish of Mannacan is.
|
Interesting Videos
These video shows the importance of Titanium and how it plays a big role in our society with it's qualities.
For more information
Bibliography
Works Cited
Bentor, Yinon. "Chemical Elements.com - Titanium (Ti)." Chemical Elements.com - Titanium (Ti). Yinon Bentor, 2012. Web. 19 Mar. 2013.
Hsu, David D. "Titanium Element Facts." Chemicool. David D Hsu, 1996. Web. 15 Mar. 2013.
Lagasse, Paul. "Titanium." The Columbia Encyclopedia. 6th ed. N.p.: Columbia UP, 2000. Http://infotrac.galegroup.com. Web. 17 Mar. 2013.
Stwertka, Albert. A Guide to the Elements. 3rd ed. New York: Oxford UP, 2002. Print.
Bentor, Yinon. "Chemical Elements.com - Titanium (Ti)." Chemical Elements.com - Titanium (Ti). Yinon Bentor, 2012. Web. 19 Mar. 2013.
Hsu, David D. "Titanium Element Facts." Chemicool. David D Hsu, 1996. Web. 15 Mar. 2013.
Lagasse, Paul. "Titanium." The Columbia Encyclopedia. 6th ed. N.p.: Columbia UP, 2000. Http://infotrac.galegroup.com. Web. 17 Mar. 2013.
Stwertka, Albert. A Guide to the Elements. 3rd ed. New York: Oxford UP, 2002. Print.