Aluminum
http://periodictable.com/Samples/013.21/s9s.JPG
Before we learn about aluminum we need to know what it is
What is Aluminum?
Aluminum is a silvery, light, soft, solid element(at it's pure form)! Aluminum is in the group metal.
Chemical name: Aluminum
Chemical symbol: Al
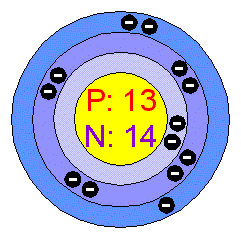
Atomic number: 13
Atomic mass: 26.98
Protons(positive charge):13
Neutrons(no charge): 14(Atomic mass rounded: 27 than subtract protons or electrons=14)
Electrons(negative charge):13
Why is ot important: It is versatile(Has many uses) Without it we might not have the things we have!
Element group name: Metal!
Color: silver- white
Melting point:1220.58°F(rounded)
boiling point: 4566°F
What has aluminum in it?
Many things have aluminum here are some.
-Cans
-Wind chimes
-Ladders
-Computers
-Phones
-airplanes
-Aluminum foil
-Scooters
Aluminum is most likely to be found in compounds(usually found with oxygen)
Aluminum is a good conductor of electricity.
Melted Aluminum: http://www.mansellandassociates.net/RotaryFurnacepour1.JPG.JPG
Videos all about aluminum:

Who discovered aluminum?
It was Hans Christian Orsted! He isolated it in 1825. He discovered aluminum by "reducing aluminum chloride using a potassium-mercury amalgam*:. Aluminum the element was discovered in Copenhagen, Denmark * Amalgam means: "an alloy of mercury with another metal or metals." Alloy:a" substance composed of two or more metals" Both defintions provided by dictionary.com
http://www.magnet.fsu.edu/education/tutorials/pioneers/images/hanschristianoersted.jpg
It was Hans Christian Orsted! He isolated it in 1825. He discovered aluminum by "reducing aluminum chloride using a potassium-mercury amalgam*:. Aluminum the element was discovered in Copenhagen, Denmark * Amalgam means: "an alloy of mercury with another metal or metals." Alloy:a" substance composed of two or more metals" Both defintions provided by dictionary.com
http://www.magnet.fsu.edu/education/tutorials/pioneers/images/hanschristianoersted.jpg
Did you know?
In ancient times, aluminum was used for dyeing, tanning
and for stopping bleeding.
The half life of aluminum is 730,000 years!!
Many years ago aluminum was more precious than gold!
3.4 million tons of aluminum went to U.S. solid waste stream.
In ancient times, aluminum was used for dyeing, tanning
and for stopping bleeding.
The half life of aluminum is 730,000 years!!
Many years ago aluminum was more precious than gold!
3.4 million tons of aluminum went to U.S. solid waste stream.
Works Cited
"Aluminum." Http://science.marshall.edu/castella/chm448/elements1.pdf. N.p., n.d. Web.
"Aluminum." InfoTrac Junior Edition. Gale, n.d. Web. 19 Mar. 2013. <http://infotrac.galegroup.com/k12/infomark/326/858/3925772w16/purl=rc1_K12J_
0_A69013801&dyn=4!xrn_2_0_A69013801?sw_aep=mlin_m_big>.
Bentor, Yinon. "Chemical Elements.com - An Interactive Periodic Table of the Elements." Chemical Elements.com - An Interactive Periodic Table of the Elements. N.p., Feb. 1996. Web. 24 Mar. 2013.
Hsu, David D. "The Periodic Table." Periodic Table of Elements and Chemistry. N.p., 1996. Web. 24 Mar. 2013.
Stwertka, Albert. "Aluminum." A Guide to the Elements. New York: Oxford UP, 2002. N. pag. Print.