Xenon Xe
http://periodictable.com/Samples/054.6/s9s.JPG
History and Physical Features
Xenon was discovered in July 12, 1898 by Sir William Ramsay and Morris M. Travers. It is a colorless, very heavy , odorless noble gas. It can be found after evaporating liquid usually oxygen and fluorine. It's a Solid and the element group is Non Metal. Its boiling point is -162.58 °F and its melting point is -169.42 °F . Xenon is part of a Greek word called Xenos meaning strange and Xenon means stranger.
http://www.chemicalelements.com/bohr/b0054.gif
Why is Xenon Important?
Xenon is important because it can be used as a light source, stroboscopes,high speed photography, and medicine.
Uses of Xenon
It can be used as a lasers, atomic energy field in bubble chambers, and making electron tubes.
http://tuningautogroup.com/images/xenonhalogen2.jpg
Facts about Xenon
What is the atomic weight and number? 131.29 and 54
Where was it found? United Kingdom in Europe
Is it toxic? No but some compounds are toxic.
What is the boiling point of Xenon? -162.6F
What is the Melting point of Xenon? 169.42F
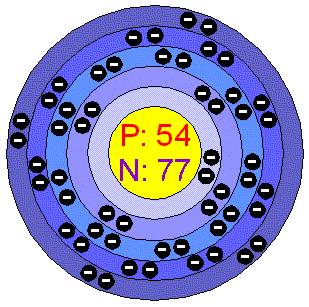
How much Protons/Electrons does it have? 54
How many Neutrons does it have? 77
What does noble gas mean? It means the gas can't be reacted in a chemical way.
Fun fact! 0.0000087% of the Earth's atmosphere is Xenon
Fun fact! When Xenon is inhaled it can make your voice deeper because it is denser then helium.
What is the atomic weight and number? 131.29 and 54
Where was it found? United Kingdom in Europe
Is it toxic? No but some compounds are toxic.
What is the boiling point of Xenon? -162.6F
What is the Melting point of Xenon? 169.42F
How much Protons/Electrons does it have? 54
How many Neutrons does it have? 77
What does noble gas mean? It means the gas can't be reacted in a chemical way.
Fun fact! 0.0000087% of the Earth's atmosphere is Xenon
Fun fact! When Xenon is inhaled it can make your voice deeper because it is denser then helium.
Bibliography
Sites:
"Xenon Facts." About.com Chemistry. IAC/InterActiveCorp, 1996. Web. 24 Mar. 2013.
Stewart, Doug.
"Xenon Element Facts." Chemicool. Doug Stewart, 1996. Web. 24 Mar. 2013. Bentor, Yinon. "Xenon (Xe)." Chemical Elements.com - Xenon (Xe). Yinon Bentor, 1996. Web. 24 Mar. 2013. Gagnon, Steve. "The Element Xenon." It's Elemental -. Steve Gagnon, n.d. Web. 24 Mar. 2013
Book and Encylopedia :
The World Book Encyclopedia. Chicago: World Book, 2007. Print. Schrobilgen, Gary J. "Xenon." Britannica School. Encyclopædia Britannica, Inc.,
2013. Web. 27 Mar. 2013.
"Xenon Facts." About.com Chemistry. IAC/InterActiveCorp, 1996. Web. 24 Mar. 2013.
Stewart, Doug.
"Xenon Element Facts." Chemicool. Doug Stewart, 1996. Web. 24 Mar. 2013. Bentor, Yinon. "Xenon (Xe)." Chemical Elements.com - Xenon (Xe). Yinon Bentor, 1996. Web. 24 Mar. 2013. Gagnon, Steve. "The Element Xenon." It's Elemental -. Steve Gagnon, n.d. Web. 24 Mar. 2013
Book and Encylopedia :
The World Book Encyclopedia. Chicago: World Book, 2007. Print. Schrobilgen, Gary J. "Xenon." Britannica School. Encyclopædia Britannica, Inc.,
2013. Web. 27 Mar. 2013.