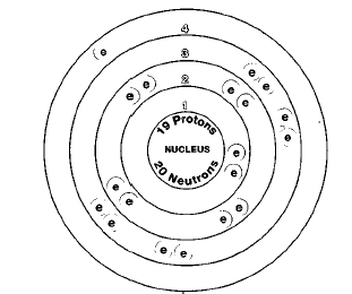
Potassium k

Atomic Number : 19
Atomic Mass: 39.10
Part of matter: solid
Color: silver, metallic
Element Group Name: Metal
Specific Gravity: 0.8
Atomic weight: 39.0983 (1)
http://img.ehowcdn.com/article-new/ehow/images/a08/3v/9s/type-element-potassium-800x800.jpg
Atomic Mass: 39.10
Part of matter: solid
Color: silver, metallic
Element Group Name: Metal
Specific Gravity: 0.8
Atomic weight: 39.0983 (1)
http://img.ehowcdn.com/article-new/ehow/images/a08/3v/9s/type-element-potassium-800x800.jpg
How Potassium was discovered
Earlier in time people had something called potash, a compound of potassium that is made when wood burns. When you wash the wood ash with water the potash dissolves. Vegetable alkali is the same thing as potash. Potassium is the chemical name for potash.
There is also is a substance called mineral alkali. Its made by different types of rocks. Mineral alkali was often called soda ash. Now people call it sodium carbonate.
For a while during the years people often got "vegetable alkali" and "mineral alkali" confused. They both acted very much alike, such as they both could be used for cleaning. Scientist finally, in the eighteenth century, understood that they came from different sources.
In the late 1700s, chemist found out they both contained an element they never found before. They tried to break down the compound into a single element. The first way Davy tried was to put an electrical current into a water solution of one compound or the other. Davy didn't realized that sodium and potassium are active. When you do that the electrical current is freed.
Davy realized that if you melt a little of potash and soda ash, then pass it through an electrical current, you will see a tiny liquid droplet of metal formed in each case. When Davy did this it was th first time humans ever saw Potassium and Sodium.
When using this process, Davy first succeeded on October 6, 1807. After a few day Davy tried again but with soda ash and got pure sodium metal.
Davy named these two elements after their much older names: potassium for "potash" and sodium for "soda ash.
Earlier in time people had something called potash, a compound of potassium that is made when wood burns. When you wash the wood ash with water the potash dissolves. Vegetable alkali is the same thing as potash. Potassium is the chemical name for potash.
There is also is a substance called mineral alkali. Its made by different types of rocks. Mineral alkali was often called soda ash. Now people call it sodium carbonate.
For a while during the years people often got "vegetable alkali" and "mineral alkali" confused. They both acted very much alike, such as they both could be used for cleaning. Scientist finally, in the eighteenth century, understood that they came from different sources.
In the late 1700s, chemist found out they both contained an element they never found before. They tried to break down the compound into a single element. The first way Davy tried was to put an electrical current into a water solution of one compound or the other. Davy didn't realized that sodium and potassium are active. When you do that the electrical current is freed.
Davy realized that if you melt a little of potash and soda ash, then pass it through an electrical current, you will see a tiny liquid droplet of metal formed in each case. When Davy did this it was th first time humans ever saw Potassium and Sodium.
When using this process, Davy first succeeded on October 6, 1807. After a few day Davy tried again but with soda ash and got pure sodium metal.
Davy named these two elements after their much older names: potassium for "potash" and sodium for "soda ash.
Why is Potassium Important?
Potassium is important for many reasons. One reason is that it helps our metabolism. Our metabolism is what makes food into energy. Another reason is that it helps enzymes( somespeed up some chemical reactions in the liver and the muscles.
http://www.drwhitaker.com/resources/drwhitaker/images/article/potassium_220.jpg

Where to find this element and how is it used?
- bananas
- oranges
- 2.5 of the earths crust
- compounds in the dead sea
- metals
- metabolism
- Medicine *
- fertilizer
- killiing weeds
- matches (saltpeter)
How do you say it?po·tas·si·um
Physical Properties
- Potassium is one of the most reactive and electropositive (electrically positive) of metals
- The metal must be stored in a mineral oil, such as kerosene, as it oxidizes rapidly in air and catches fire spontaneously when exposed to water.
- Potassium and its salts will color flames violet
- soft, waxy, silvery-white metal
- Melting Point: 146.08 °F 63.38°c 336.53 k
- Boiling point:1398 °F 759°c 1,032.15 k
6 interesting facts:
- Potassium is the second lightest metal (first being lithium) So soft you can cut it with a knife!!
- Cooling Point: 146°f Melting Point: 146.08 °F Boiling point:1398 °F
- Potassium is one of the most reactive and electropositive (electrically positive) of metals
- The metal must be stored in a mineral oil, such as kerosene, as it oxidizes rapidly in air and catches fire spontaneously when exposed to water. Its decomposition in water evolves hydrogen
- Potassium and its salts will color flames violet
- soft, waxy, silvery-white metal
Sources
Gangon, Steve. "The Element Potassium." It's Elemental. Steve Ganon, 17 Mar. 2013. Web. 17 Mar. 2013.
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. Print.
Network, Answering Publishing. "What Is the Melting Point for Potassium?" WikiAnswers. Answers, 2013. Web. 22 Mar. 2013.
Shriver, Duward F. "Potassium." The World Book Encyclopedia. Chicago, IL: World Book, 2009. N. pag. Print.
Winter, Mark. "Potassium." WebElements Periodic Table of the Elements. Webelements, 2012. Web. 17 Mar. 2013.
Gangon, Steve. "The Element Potassium." It's Elemental. Steve Ganon, 17 Mar. 2013. Web. 17 Mar. 2013.
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. Print.
Network, Answering Publishing. "What Is the Melting Point for Potassium?" WikiAnswers. Answers, 2013. Web. 22 Mar. 2013.
Shriver, Duward F. "Potassium." The World Book Encyclopedia. Chicago, IL: World Book, 2009. N. pag. Print.
Winter, Mark. "Potassium." WebElements Periodic Table of the Elements. Webelements, 2012. Web. 17 Mar. 2013.