http://www.nist.gov/pml/data/platinum/images/shutterstock_24397567_fzd-it_2.jpg
Platinum was discovered by south american people they used it to make artifacts.Some things that have or involve platinum in them are:rings,bracelets,crowns,neckleses ,technology,wires experiments,and coins.Platinum is used in catalytic converters.Catalytic converters combine carbon monoxide and unburned fuel from the cars exhaust with oxygen making Co2 and water vapor.I think platinum is important because its often used in jewelry and its rarer than gold.
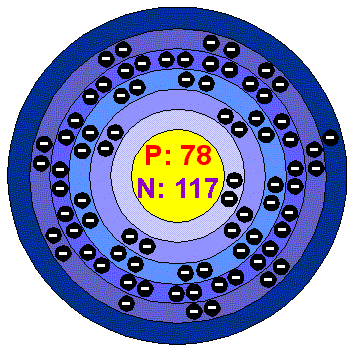
Platinum's atomic number is 78
platinum's element symbol is Pt
platinum's element name is platinum
platinum's atomic mass is 195.084
Platinum's group name is (metal)
Platinum's melting point 3215.1°F
Platinum's boiling point 6917°F
Platinum's density is 21.46
Platinum's color grayish white
Fun facts
Platinum is found in 1 out of 5 devices.
Platinum wasn't noticed by western scientist until 1735.
Platinum is rarer than gold
Platinum is more durable than gold
Platinum is very resistant to acids.
Platinum is used in catalytic converters.Catalytic converters combine carbon monoxide and unburned fuel from the cars exhaust with oxygen making Co2 and water vapor.
This is a chart comparing Platinum to Gold
|
Platinum
Platinum's atomic number is 78 platinum's element symbol is Pt platinum's element name is platinum platinum's atomic mass is 195.084 Platinum's group name is metal Platinum's melting point 3215.1°F Platinum's boiling point 6917°F Platinum's density is 21.46 |
Gold
Gold's atomic number is 79 Gold's element symbol is Au Gold's element name is gold Gold's atomic mass is 196.966569 Gold's group name is metal Gold's melting point is 1947.52°F Gold's boiling point is 5173°F Gold's density is 19.282 |
http://www.chemicalelements.com/bohr/b0078.gif
Bibliography
Ganon, Steve. "The Element Platinum." It's Elemental -. N.p., n.d. Web. 25 Mar. 2013.
McKeever, Susan, ed. The DK Science Encyclopedia. New York: DK Pub., 1994. Print.
Stwertka, Albert. A Guide to the Elements. New York: Oxford UP, 1998. Print.
McKeever, Susan, ed. The DK Science Encyclopedia. New York: DK Pub., 1994. Print.
Stwertka, Albert. A Guide to the Elements. New York: Oxford UP, 1998. Print.
http://us.123rf.com/400wm/400/400/dvarg/dvarg1205/dvarg120500007/13502276-green-atom-electron-llustration-on-black-background.jpg