KRYPTON (KR)
By: Matt Geis
Krypton was discovered by a scientist named William Ramsay and his student Morris Travers

Ramsay and Travers discovered it on May 30th, 1898 while trying to isolate neon. Isolate means: to cause (a person or place) to be or remain alone or apart from others. Back in that time neither neon or Krypton had been discovered. It was named krypton because that meant "hidden" in Greek because when they discovered it they only knew it was there because there was a reaction that made it's yellow and green colors appear. Krypton isn't very important because it isn't used in very many scientific experiments and there are very little objects that use krypton in it.
Where Krypton was discovered (United Kingdom)
http://www.nature.com/news/2003/030813/images/alpha_180.jpg
Atomic structure
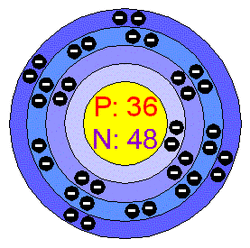
The picture on the left shows the Atomic Structure. This Atomic Structure has four levels
First Energy level: 2
Second Energy Level: 8
Third Energy Level: 18
Fourth Energy Level: 8
Energy levels: The fixed amount of energy that a system described by quantum mechanics, such as a molecule, atom, electron, or nucleus, can have.
http://www.chemicalelements.com/bohr/b0036.gif
First Energy level: 2
Second Energy Level: 8
Third Energy Level: 18
Fourth Energy Level: 8
Energy levels: The fixed amount of energy that a system described by quantum mechanics, such as a molecule, atom, electron, or nucleus, can have.
http://www.chemicalelements.com/bohr/b0036.gif
The Facts
Atomic mass: 83.80
Number of electrons and protons in the nucleus (atomic number): 36
Number of neutrons: 48
Original state: Gas
Element group name: Nonmetal
boiling temperature: -153.2 degrees Celsius
freezing temperature: -156.6 degrees Celsius
Half life: 10.756 seconds
Name Origin: from the Greek word kryptos (hidden)
Number of electrons and protons in the nucleus (atomic number): 36
Number of neutrons: 48
Original state: Gas
Element group name: Nonmetal
boiling temperature: -153.2 degrees Celsius
freezing temperature: -156.6 degrees Celsius
Half life: 10.756 seconds
Name Origin: from the Greek word kryptos (hidden)
Where is it used?
1) Small traces found in Earth's Atmosphere
2) Fluorescent lamps
3) Neon lighting
4) Some types of lasers
5) Electric tubes
6) The space in between double spaced window panes
7) Flash photography
2) Fluorescent lamps
3) Neon lighting
4) Some types of lasers
5) Electric tubes
6) The space in between double spaced window panes
7) Flash photography
Krypton laser
http://www.youtube.com/watch?v=gJdf9pfS9D0
Cool Facts

1) it is famously known as superman's only weakness.
2) In it's original form krypton is Colorless, Odorless, and Tasteless.
3) The cost of one gallon of krypton is $11.56.
4) Belongs to the group of noble gasses along with Neon, Argon, Xenon, and Radon.
5) The discoverers of Krypton (Ramsay and Tavers) also discovered xenon and neon.
2) In it's original form krypton is Colorless, Odorless, and Tasteless.
3) The cost of one gallon of krypton is $11.56.
4) Belongs to the group of noble gasses along with Neon, Argon, Xenon, and Radon.
5) The discoverers of Krypton (Ramsay and Tavers) also discovered xenon and neon.
http://images2.wikia.nocookie.net/__cb20100521192034/exodus3000/images/9/9c/Krypton-glow.jpg
Like Krypton after reading my page?
Take my quiz!
Were you correct?

Answers:
1) A
2) C
3) C
http://www.chrishatch.org/wp-content/uploads/2013/02/100-percent.jpg
1) A
2) C
3) C
http://www.chrishatch.org/wp-content/uploads/2013/02/100-percent.jpg
Bibliography
Works Cited
Andrew Radar Studios. "Krypt-on." CHEM4KIDS. Andrew Radar Studios, 1997. Web. 24 Mar. 2013.
D. Hsu. of the Massachusetts Institute of Technology, David. "The Periodic Table." Periodic Table of Elements and Chemistry. David D. Hsu. of the Massachusetts Institute of Technology, 2013. Web. 24 Mar. 2013.
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. Print.
The World Book Encyclopedia. Chicago, IL: World Book, 2009. Print.
Andrew Radar Studios. "Krypt-on." CHEM4KIDS. Andrew Radar Studios, 1997. Web. 24 Mar. 2013.
D. Hsu. of the Massachusetts Institute of Technology, David. "The Periodic Table." Periodic Table of Elements and Chemistry. David D. Hsu. of the Massachusetts Institute of Technology, 2013. Web. 24 Mar. 2013.
Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. Print.
The World Book Encyclopedia. Chicago, IL: World Book, 2009. Print.
