Hydrogen (H)
by:Tina Huang
**Pronounced: High-Droe-Ginn
Discoveries
Hydrogen is a gas that was discovered in 1766, by Henry Cavendish. Hydrogen is a colorless, odorless, tasteless gas. Hydrogen is considered important because hydrogen can be used to carry energy from its source to the place where it is required. Hydrogen is the lightest gas and most of hydrogen is found in water, very little found in fuel because of its light-weight.
|
Uses of Hydrogen:
1. "Town Gas" was a gas that many people in the towns use. It has a mixture of hydrogen, carbon monoxide, carbon dioxide and methane. 2. Synthetic fertilizer. This was used to help farmers grow large crops easier. 3. Water. 4. Hydrogen fuel. Hydrogen can make fuel for people. 5. Hot air balloons. 6. Delivery trucks(that are hydrogen powered). 7. Hydrogen energy. 8. Hydrogen gas. 9. Hydrogen power. 10. Air. When hydrogen is in air, it is combined with oxygen and many other elements and particles. |
Interesting Hydrogen Rap
Uses:
The most important single use of hydrogen is in the manufacture of ammonia. Ammonia is made combining hydrogen and nitrogen at high pressure and temperature in the presence of a catalyst. A catalyst is a substance used to speed up or slow down a chemical reaction. Ammonia is a very important compound. The most important product is fertilizer.
More information:

http://upload.wikimedia.org/wikipedia/commons/9/9c/HydrogenBox.png
Atomic Number: 1
Atomic Weight: 1.00794
Element Symbol: H
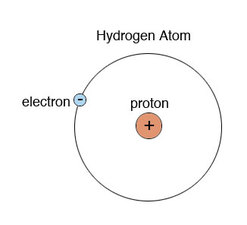
Number of Protons: 1
Number of Electrons: 1
Number of Neutrons: 0
Melting Point: 13.81 K (-259.34°C or -434.81°F)
Boiling Point: 20.28 K (-252.87°C or -423.17°F)
Density: 0.00008988 grams per cubic centimeter
Phase at Room Temperature: Gas
Element Classification: Non-metal
Color: Colorless
Atomic Weight: 1.00794
Element Symbol: H
Number of Protons: 1
Number of Electrons: 1
Number of Neutrons: 0
Melting Point: 13.81 K (-259.34°C or -434.81°F)
Boiling Point: 20.28 K (-252.87°C or -423.17°F)
Density: 0.00008988 grams per cubic centimeter
Phase at Room Temperature: Gas
Element Classification: Non-metal
Color: Colorless
Interesting Facts:
1. New hydrogen discoveries and uses are constantly being found everyday.
2. The same inflammable air that had been studied for more than 100 years by Robert Boyle and other scientists.
3. Hydrogen is one of the the most abundant elements in the universe. Nearly 9 out of 10 atoms in the universe are hydrogen atoms.
4. Hydrogen is also common in Earth. It is the third most abundant element after oxygen and silicon. About 15% of the atoms on Earth are hydrogen atoms.
5. Hydrogen was named by a French chemist Antonie-Laurent Lavoiser.
2. The same inflammable air that had been studied for more than 100 years by Robert Boyle and other scientists.
3. Hydrogen is one of the the most abundant elements in the universe. Nearly 9 out of 10 atoms in the universe are hydrogen atoms.
4. Hydrogen is also common in Earth. It is the third most abundant element after oxygen and silicon. About 15% of the atoms on Earth are hydrogen atoms.
5. Hydrogen was named by a French chemist Antonie-Laurent Lavoiser.
Bibliography
Works Cited
Bentor, Yinon. "Chemical Elements.com - Hydrogen (H)." Chemical Elements.com - Hydrogen (H). N.p., n.d. Web. 15 Mar. 2013.
Farndon, John. Hydrogen. New York: Benchmark, 2000. Print.
"Hydrogen." InfoTrac Student Edition. Gale, 2000. Web. 17 Mar. 2013.
Studios, Andrew Rador. "Where Else Can You Find Hydrogen?" Chem4Kids.com: Hydrogen: Say It. N.p., 2012. Web. 17 Mar. 2013.
Bentor, Yinon. "Chemical Elements.com - Hydrogen (H)." Chemical Elements.com - Hydrogen (H). N.p., n.d. Web. 15 Mar. 2013.
Farndon, John. Hydrogen. New York: Benchmark, 2000. Print.
"Hydrogen." InfoTrac Student Edition. Gale, 2000. Web. 17 Mar. 2013.
Studios, Andrew Rador. "Where Else Can You Find Hydrogen?" Chem4Kids.com: Hydrogen: Say It. N.p., 2012. Web. 17 Mar. 2013.