Germanium Ge
Building Blocks
*Chemical name: germanium
*Chemical symbol:Ge
*Atomic number:32
*Atomic mass: 72.61
*Solid, liquid, gas, or synthetic?:solid
*Color:gray or white
*Element group name: metaloid
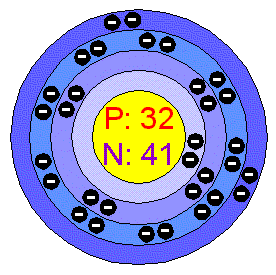
*Number of electrons:about 32
*Number of protons:about 32
*Number of neutrons:about 42
*Pronunciation :jer-MAY-nee-um
*Chemical name: germanium
*Chemical symbol:Ge
*Atomic number:32
*Atomic mass: 72.61
*Solid, liquid, gas, or synthetic?:solid
*Color:gray or white
*Element group name: metaloid
*Number of electrons:about 32
*Number of protons:about 32
*Number of neutrons:about 42
*Pronunciation :jer-MAY-nee-um
Discovery
In the 1860's, Mendeleev decided he wanted to order the periodic table of elements by atomic number, but he found that the element with the atomic number 32 was missing. Mendeleev knew that this was nothing to worry about. element 32 had not been discovered, that was all. Germanium was descovered in 1885, by the scientist Clements Alexander. Germanium was discovered neer Freiburg, Germany (around where silver and sulfer were discovered) when Alexander noticed that aragonite was only 94% mass. Clements Alexander named his new found element "germanium", in honor of his home country.
Physical Properties
Germanium can be gray, white or grayish-white. It is brittle, lustery, and partially metallic. It is not a good conductor, instead, it is used as a semi-conductor. It is metalloid.
Uses
Germanium is used in solar cells. That makes it important because it helps make solar panels, witch can be used to collect solar(radiant) energy. Other uses are solar panels, iPods, smart phones, Television, transistors, circuits and as a semiconductor and an alloying agent.
In the 1860's, Mendeleev decided he wanted to order the periodic table of elements by atomic number, but he found that the element with the atomic number 32 was missing. Mendeleev knew that this was nothing to worry about. element 32 had not been discovered, that was all. Germanium was descovered in 1885, by the scientist Clements Alexander. Germanium was discovered neer Freiburg, Germany (around where silver and sulfer were discovered) when Alexander noticed that aragonite was only 94% mass. Clements Alexander named his new found element "germanium", in honor of his home country.
Physical Properties
Germanium can be gray, white or grayish-white. It is brittle, lustery, and partially metallic. It is not a good conductor, instead, it is used as a semi-conductor. It is metalloid.
Uses
Germanium is used in solar cells. That makes it important because it helps make solar panels, witch can be used to collect solar(radiant) energy. Other uses are solar panels, iPods, smart phones, Television, transistors, circuits and as a semiconductor and an alloying agent.
Fun Facts
*Germanium's boiling point is 2830 degrees C
*Germanium's electron configuration is [Ar](3d)10(4s)2(4p)2
*Germanium's electron shells are 2,8,18 and 4
*Germanium is toxic
*Germanium's boiling point is 2830 degrees C
*Germanium's electron configuration is [Ar](3d)10(4s)2(4p)2
*Germanium's electron shells are 2,8,18 and 4
*Germanium is toxic
Bibliography
Dingle, Adrian. The Periodic Table: Elements with Style. New York: Kingfisher, 2007. Print.
"Germanium." Wikipedia. Wikimedia Foundation, 25 Mar. 2013. Web. 24 Mar. 2013.
Stewart, Doug Stewart. "Germanium Element Facts." Chemicool. N.p., n.d. Web. 15 Mar. 2013.
Dingle, Adrian. The Periodic Table: Elements with Style. New York: Kingfisher, 2007. Print.
"Germanium." Wikipedia. Wikimedia Foundation, 25 Mar. 2013. Web. 24 Mar. 2013.
Stewart, Doug Stewart. "Germanium Element Facts." Chemicool. N.p., n.d. Web. 15 Mar. 2013.