Silicon
http://periodictable.com/Samples/014.12/s9s.JPG
Overviewing Video
http://www.youtube.com/watch?v=usA86CLZmb4
General Info.-Basics
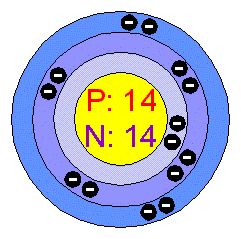
Chemical name: Silicon
Chemical symbol: Si
Atomic number: 14
Atomic mass: 28.086
Solid, liquid, gas or synthetic: Solid
Color: Grey
Element group name: Metalloid
Melting Point: 1410.0 °C (2570.0 °F)
Boiling Point: 2355.0 °C (4271.0 °F)
Crystal Structure: Cubic
Isotopes: 14
Stable isotopes: 3
Half lives: 100 or 2.62 hours
Density @ 20 degrees C: 2.33 g/cm3
3p2g/cm3http://chemicalelements.com/bohr/b0014.gifhttp://chemicalelements.com/bohr/b0014.gif
Chemical symbol: Si
Atomic number: 14
Atomic mass: 28.086
Solid, liquid, gas or synthetic: Solid
Color: Grey
Element group name: Metalloid
Melting Point: 1410.0 °C (2570.0 °F)
Boiling Point: 2355.0 °C (4271.0 °F)
Crystal Structure: Cubic
Isotopes: 14
Stable isotopes: 3
Half lives: 100 or 2.62 hours
Density @ 20 degrees C: 2.33 g/cm3
3p2g/cm3http://chemicalelements.com/bohr/b0014.gifhttp://chemicalelements.com/bohr/b0014.gif
Discovery and Uses
|
Jons Jakob Berzelius discovered the hard, grey element that was silicon in 1824. He did this by heating potassium chips in a silica container, then he washed away all extra residue and repeated the process.
|
The element silicon has a lot of uses, and it is extremely important because of that. A example of this would be that silicon is the second most abundant element in the earths crust, and this planet might not exist without it. Other uses of silicon are:
1.) Quartz 2.) Flint 3.) The last stage of a heavy star 4.) Silicon chips 5.) Sand 6.) Granite 7.) Ceramics 8.) Your skeleton 9.) Silicone 10.) Medical implants 11.) Enhancing irons magnetic field 12.) Engines 13.)Abrasives 14.) Concrete 15.) Bricks 16.) Precise watches and clocks 17.) Glass 18.) Cookware 19.) Sealants 20.) Adhesives 21.) Insulation |
Interesting facts
1.) Silicon is the eighth most abundant element in the Universe.
2.) Before a heavy star turns Supernova, it is compromised of burning silicon.
3.) You can buy 100g of silicon for $5.4.
4.) Solid silicon does not react with most acids.
5.) Silicon expands when it freezes.
2.) Before a heavy star turns Supernova, it is compromised of burning silicon.
3.) You can buy 100g of silicon for $5.4.
4.) Solid silicon does not react with most acids.
5.) Silicon expands when it freezes.
Bibliography
Works Cited
Websites- Benter, Yinon. "Chemical Elements.com - Silicon (Si)." Chemical Elements.com - Silicon (Si). Yinon Benter, n.d. Web. 2012.
Hsu., David D. "Silicon Element Facts." Chemicool. N.p., 2013. Web. 13 Mar. 2013.
Encyclopedia- Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. 30-31. Print.
Book- McGowen, Tom. Chemistry, the Birth of a Science. New York: F. Watts, 1989. 83. Print.
Websites- Benter, Yinon. "Chemical Elements.com - Silicon (Si)." Chemical Elements.com - Silicon (Si). Yinon Benter, n.d. Web. 2012.
Hsu., David D. "Silicon Element Facts." Chemicool. N.p., 2013. Web. 13 Mar. 2013.
Encyclopedia- Knapp, Brian J., David Woodroffe, and David A. Hardy. Elements. Danbury, CT: Grolier Educational, 1996. 30-31. Print.
Book- McGowen, Tom. Chemistry, the Birth of a Science. New York: F. Watts, 1989. 83. Print.
