CURIUM (Cm)
http://images-of-elements.com/s/curium.jpg
Properties and features of Curium:
-Solid, liquid, gas, or synthetic? Synthetic
-Radioactive? Yes
-Element symbol: Cm
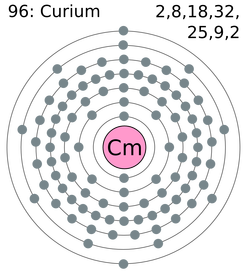
-Electron shell: 2, 8, 18, 32, 25, 9, 2 (picture to left)
-Atomic mass: 247.07
-Atomic number: 96 (electrons and protons)
-Group: Actinide
-Color: Silvery white
-Hardness: Malleable
-Melting Point: 2453 degrees Fahrenheit
-Freezing point: unknown
-Density: 13.51 grams per cubic centimeter
-Half-life: 162.8
-Most stable isotope: Curium 247
-Electrons and protons: 96
-Neutrons: 151
http://upload.wikimedia.org/wikipedia/commons/d/d8/Electron_shell_096_curium.png
-Radioactive? Yes
-Element symbol: Cm
-Electron shell: 2, 8, 18, 32, 25, 9, 2 (picture to left)
-Atomic mass: 247.07
-Atomic number: 96 (electrons and protons)
-Group: Actinide
-Color: Silvery white
-Hardness: Malleable
-Melting Point: 2453 degrees Fahrenheit
-Freezing point: unknown
-Density: 13.51 grams per cubic centimeter
-Half-life: 162.8
-Most stable isotope: Curium 247
-Electrons and protons: 96
-Neutrons: 151
http://upload.wikimedia.org/wikipedia/commons/d/d8/Electron_shell_096_curium.png
http://www.youtube.com/watch?feature=player_detailpage&v=0__NPdVBlm0
How was curium discovered?
Curium was discovered by Glenn T. Seaborg, Ralph A. James and Albert Ghiorso in July, 1994. They bombarded plutonium with tiny subatomic particles called alpha particles in a device called a cyclotron. The alpha particle adds two protons to the nucleus, which explains the jump from plutonium, which is number 94, to curium, which is number 96.
Curium was discovered by Glenn T. Seaborg, Ralph A. James and Albert Ghiorso in July, 1994. They bombarded plutonium with tiny subatomic particles called alpha particles in a device called a cyclotron. The alpha particle adds two protons to the nucleus, which explains the jump from plutonium, which is number 94, to curium, which is number 96.
How was curium given it's name?
Curium is similar to gadolinium, which was named after Johan Gadolin, an expert in rare-earth chemistry. Because of this similarity, Curium was named after a pair famous scientists, Pierre and Marie Curie.
Curium is similar to gadolinium, which was named after Johan Gadolin, an expert in rare-earth chemistry. Because of this similarity, Curium was named after a pair famous scientists, Pierre and Marie Curie.
Where is Curium used?

Curium is synthetic, so it is not used in many places, but here are some places that it could be used:
1.) In an alpha proton x-ray spectrometer on the Mars Rover which determines what elements are in rock (Picture to the left)
2.) Thermoelectric power source
3.) For research in a lab
http://mars.jpl.nasa.gov/msl/images/apxs_portrait.jpg
1.) In an alpha proton x-ray spectrometer on the Mars Rover which determines what elements are in rock (Picture to the left)
2.) Thermoelectric power source
3.) For research in a lab
http://mars.jpl.nasa.gov/msl/images/apxs_portrait.jpg
Curium Isotopes:
|
Mass Number:
233 234 235 236 237 238 239 240 241 242 243 244 245 246 247 248 249 250 251 252 |
Half-Life:
23 51 N/A N/A N/A 2.4 Hours about 2.9 hours 27 days 32.8 days 162.8 days 29.1 Years 18.1 years 8423 years 4706 years 15,600,000 years 348000 years 64.15 minutes N/A 16.8 minutes less than 2 days |
Cool facts about curium:
1.) Curium is so radioactive that it glows in the dark! Energy from decaying curium is what makes this happen.
2.) The reason Curium is not used as a thermoelectric power source is because it is so radioactive. It stays in bones and destroys the marrow, which is a main producer of red blood cells in your body, so basically, curium destroys red blood cells.
3.) Curium loses color gradually in dry air at room temperature.
4.) Only several kilograms of curium are produced each year.
Here are some websites I found that were helpful:
Bibliography:
Internet Sources:
1.) The Element Curium, It’s Elemental, http://education.jlab.org/itselemental/ele096.html 3-12-13
2.) Curium, Visual Elements Periodic Table, http://www.rsc.org/periodic-table, 3-12-13
3.) Curium: uses, WebElements, http://www.webelements.com/, 3-12-13
4.) Curium Element Facts, Chemicool, www.chemicool.com, 3-12-13
5.) Curium, Chemistry Explained, http://www.chemistryexplained.com/, 3-13-12
6.) Curium Periodic Table of Elements - wood board, Cutcaster, http://cutcaster.com/photo/100786101-Curium-Periodic-Table-of-Elements-wood-board/, 3-13-12
7.) Curium #96, Origin of the Element Names, http://d1068036.site.myhosting.com/people/Curium.html, 3-15-13
8.) Alpha Particle, Universe Today, http://www.universetoday.com/, 3-17-13
9.) Specific Gravity of Curium, Chemistry Data.com, http://www.noblemind.com/search.exe?keyword=Curium+Specific+Gravity&var=2, 3-17-13
10.) Element Curium - Cm, Enviromental Chemistry, http://environmentalchemistry.com/, 3-20-23
Book Sources:
1.)Asimov, Isaac,Asimov's Chronology of Science & Discovery, HarperCollins, 3-18-13
Encyclopedia Sources:
1.) Heiserman, David, Exploring Chemical Elements and their Compounds, United States of America, TAB Books, 1992
Other Sources:
1.) http://youtu.be/0__NPdVBlm0

