Titanium
(Ti)
Titanium! This is one awesome element!

Titanium is one of the most important elements we know of today. Why? Not only is its silvery-white color pleasing to the eye, but it's the ninth most commonly occurring element on the earth's crust, making it very useful for humans today.
http://pss.scdsb.on.ca/Departments/Science/SCH4U/Davis%20and%20Josh%20Titanium%20Website/titanium.jpg
http://pss.scdsb.on.ca/Departments/Science/SCH4U/Davis%20and%20Josh%20Titanium%20Website/titanium.jpg
Titanium: Uses and Purposes!
1. Propeller Shafts
2. Titanium dioxide (titania) is what 95% of titanium production is in the form of. Titania is an intensely white pigment with a strong light absorption. This form of Titanium is in the following:
a. white paint
b. food coloring
c. toothpaste
d. plastics
e. sunscreen
3. Airplanes
4. Parts of boats that are exposed to seawater
5. Missiles
6. Rockets
7. Drill bits
8. Bicycles
9. Golf clubs
10. Watches
11. Laptop computers
12. Joint replacement implants
1. Propeller Shafts
2. Titanium dioxide (titania) is what 95% of titanium production is in the form of. Titania is an intensely white pigment with a strong light absorption. This form of Titanium is in the following:
a. white paint
b. food coloring
c. toothpaste
d. plastics
e. sunscreen
3. Airplanes
4. Parts of boats that are exposed to seawater
5. Missiles
6. Rockets
7. Drill bits
8. Bicycles
9. Golf clubs
10. Watches
11. Laptop computers
12. Joint replacement implants
General Facts:
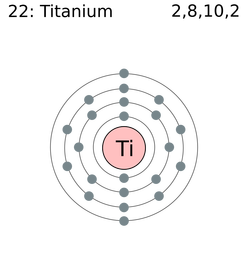
Atomic number: 22
Atomic mass: 47.87
Titanium is found as a solid in its natural form.
Titanium is silvery-white.
Titanium is a transition metal.
Melting point: 3034°F
Boiling point: 5949°F
http://upload.wikimedia.org/wikipedia/commons/c/c6/Electron_shell_022_titanium.png
Atomic mass: 47.87
Titanium is found as a solid in its natural form.
Titanium is silvery-white.
Titanium is a transition metal.
Melting point: 3034°F
Boiling point: 5949°F
http://upload.wikimedia.org/wikipedia/commons/c/c6/Electron_shell_022_titanium.png
Titanium's discovery!
Titanium was discovered by William Gregor of Cornwell, England in 1791. He found black, magnetic sand in a stream that was in the parish of Mannacan in Cornwall, England. This sand was very similar to gunpowder and contained titanium.
Titanium was discovered by William Gregor of Cornwell, England in 1791. He found black, magnetic sand in a stream that was in the parish of Mannacan in Cornwall, England. This sand was very similar to gunpowder and contained titanium.
Physical Properties:
Titanium is a silvery white transition metal. It is hard, but light, and becomes malleable when heated. It is a solid in its natural form.
Titanium is a silvery white transition metal. It is hard, but light, and becomes malleable when heated. It is a solid in its natural form.
Why I picked Titanium...

I picked Titanium because I love the song "Titanium" by David Guetta featuring Sia.
Importance: Titanium is important because it is very strong, so it is much more efficient. It is also the ninth most occurring element in the earth's crust, making it a very plentiful element. Last, the human body can not break down Titanium, which makes it very important when making joint replacement implants.
Here are the online references I used:
Fun Facts:
1. William Gregor discovered Titanium, but a man by the name of Martin Heinrich Klaproth named Titanium in the year 1795.
2. The human body can not break down titanium, so it used in hip replacements and other joint replacements.
3. Titanium is 60% heavier, but two times stronger than aluminum.
4. Titanium is 45% lighter than steel, but just as strong!
5. Titanium was named after the Titans!
6. Titanium is the ninth most abundant element in the earth's crust.
7. Titanium is the only element that will burn in an atmosphere of pure nitrogen.
1. William Gregor discovered Titanium, but a man by the name of Martin Heinrich Klaproth named Titanium in the year 1795.
2. The human body can not break down titanium, so it used in hip replacements and other joint replacements.
3. Titanium is 60% heavier, but two times stronger than aluminum.
4. Titanium is 45% lighter than steel, but just as strong!
5. Titanium was named after the Titans!
6. Titanium is the ninth most abundant element in the earth's crust.
7. Titanium is the only element that will burn in an atmosphere of pure nitrogen.
Key terms:
Pigment- a dry substance, usually reduced to particles, which can become paint, ink, etc. when suspended in a liquid vehicle.
Malleable- able to be extended or shaped. Flexible.
Pigment- a dry substance, usually reduced to particles, which can become paint, ink, etc. when suspended in a liquid vehicle.
Malleable- able to be extended or shaped. Flexible.
Works Cited
Brittanica. "Encyclopædia Britannica Online School Edition." Encyclopædia Britannica Online School Edition. Encyclopedia Britannica Online School Edition, 2013. Web. 24 Mar. 2013.
D., David. "Titanium Element Facts." Chemicool. David D. Hsu., 1996. Web. 15 Mar. 2013.
Gray, Theodore W., and Nick Mann. The Elements: A Visual Exploration of Every Known Atom in the Universe. New York: Black Dog & Leventhal, 2012. Print.
Thomas Jefferson National Accelerator Facility - Office of Science Education. "The Element Titanium." It's Elemental. Thomas Jefferson National Accelerator Facility - Office of Science Education, n.d. Web. 24 Mar. 2013.
Whitmire, Kenton H. "Titanium." World Book. T ed. Vol. 19. Chicago: World Book, 2009. 300. Print.
Brittanica. "Encyclopædia Britannica Online School Edition." Encyclopædia Britannica Online School Edition. Encyclopedia Britannica Online School Edition, 2013. Web. 24 Mar. 2013.
D., David. "Titanium Element Facts." Chemicool. David D. Hsu., 1996. Web. 15 Mar. 2013.
Gray, Theodore W., and Nick Mann. The Elements: A Visual Exploration of Every Known Atom in the Universe. New York: Black Dog & Leventhal, 2012. Print.
Thomas Jefferson National Accelerator Facility - Office of Science Education. "The Element Titanium." It's Elemental. Thomas Jefferson National Accelerator Facility - Office of Science Education, n.d. Web. 24 Mar. 2013.
Whitmire, Kenton H. "Titanium." World Book. T ed. Vol. 19. Chicago: World Book, 2009. 300. Print.
